Switching Materials: The Future of Anodes
Developing next-generation battery cells. New anode materials that combine silicon, nanophysics, and digital technologies are one of the key catalysts
Battery technology has already been proven successful on roads around the world. However, in the future, mobility may undergo a much more fundamental transformation. The electrification of powertrains has only begun. The development of battery-powered supercars, electric drones, and air cabs is just the beginning of the next chapter of this comprehensive transformation.
The lithium-ion battery is still a key technology and a significant pacesetter. Although the technology had been developed to market maturity more than three decades ago, it has continued to evolve. The technology has yet to reach its limits. However, we must continue to develop lithium-ion batteries and rethink their parts. That way, they will be able to handle the many challenges ahead as they become more widespread.
There are many approaches to this, including optimizing associated development and manufacturing processes, the accompanying use of digital technologies, and choosing the respective materials. In the coming years, for example, artificial intelligence, machine learning, and nanotechnology will help to further increase the performance of individual cells, drive innovation, and better position battery cells as decisive building blocks of an increasingly digital world. At the same time, work around innovative anode materials is progressing in leaps and bounds. As a result, all these approaches that companies, are pursuing should help to improve the efficiency of the batteries in cars and aircraft, reduce the weight and volume of batteries, and lower the cost per kilowatt-hour of storage power.

Modern lithium-ion battery cells enable a high degree of innovation through specific cell design or novel cell chemistry.
The switch to silicon offers a great opportunity for everyone
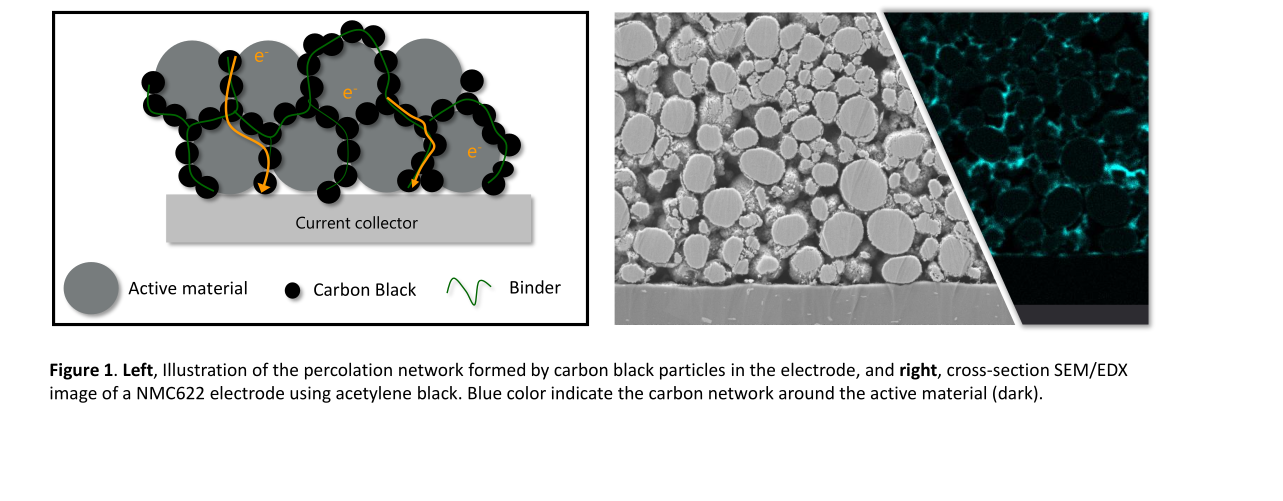
Graphite is still the most widely used active material in lithium-ion batteries – whether as natural graphite or in the form of synthetic graphite. Together with binders and conductive additives, the graphite is applied to a copper foil and serves as an anode for the intercalation of lithium atoms. However, using graphite has always been a compromise: While graphite ensures high cycle stability, it contributes significantly to the weight of the battery cell. Additionally, the theoretical specific capacity of graphite is limited to 372 milliampere hours per gram (mAh/g). In practice, it is now almost exhausted. When a holistic sustainability approach is applied to battery cell development, questions arise regarding the origin, availability, and extraction of graphite. Natural graphite is considered a critical raw material and is primarily mined in China. On the other hand, synthetic graphite is a by-product of petroleum production.
This is one of the reasons the sector is working on using alternative active materials, such as silicon. At 4,200 mAh/g, its theoretical specific capacity is ten times greater than that of graphite. As an active material in an anode, silicon would enable the construction of significantly lighter and far more powerful battery cells. Therefore, it meets certain performance parameters, especially for the further electrification of the aviation sector. On top of this, silicon is available worldwide as a raw material in almost unlimited quantities.
But theory is one thing, and practice is another. In practice, silicon is associated with considerable disadvantages that must be overcome in innovative ways. For example, silicon expands in volume by up to 300 percent during the charging process. This is significantly more than graphite, which expands by only about 10 percent. When exposed to liquid electrolytes, over time, high resistance builds up in the cell. Nevertheless, an increasing admixture of the silicon remains attractive despite these disadvantages.

Taking a nano-sponge to the silicon anode of the future
At CUSTOMCELLS, we can develop formulas for the latest materials and manufacture anodes to test them in an industry-like environment. This includes, for example, materials that contain newly structured silicon.
In recent years, the toolbox of nanotechnology has created a multitude of exciting possibilities, which our partners use and benefit us. For example, we use nanoporous silicon powder in the RoSiLIB project to develop new types of anode pastes that do not share the typical disadvantages of silicon. Even the production of this silicon powder does not require corrosive hydrofluoric acid, which is otherwise widely used to process silicon.
This is where the holistic view of sustainability comes into play. The nanoporous structure gives the silicon, which previously behaved like yeast, sponge-like properties. This means that the silicon absorbs lithium ions without expanding like silicon anodes. As a result, anodes with a silicon content of 25 percent or more are achievable and can, therefore, offer a significantly higher specific capacity without the active material comparably increasing in weight.
This approach with nanoporous material goes hand in hand with some of the other projects in which we are involved as partners. Another example is 3beLiEVe. This concerns the development of next-generation battery cells that have particularly high-performance capabilities and are completely free of critical raw materials, such as cobalt and natural graphite.
Digitization supports material development
In the future, computer simulations and reinforcement learning models can help to develop suitable electrode compositions much quicker and decisively advance the use of silicon. We are already working on these approaches in various ways. However, it is also important to emphasize, despite great progress, that the path to the fully automated development of new materials is still a long way off. This is because several materials react with each other within the cell. In addition to these interactions, a number of other boundary conditions are important to consider – from the cost-benefit ratio to scalability.
Therefore, it is still necessary to be able to test and scale materials on systems quickly to make any outcomes and interactions visible. Directly integrating sensors into cells will also help us to be able to use far more information from the proverbial wild in the future, i.e., from battery cells that are actively in use. We will, in turn, be able to draw conclusions that extend as far as material selection and composition.
The next generation of batteries is already on the way
The world is experiencing immense technological change. The issue of efficiency is becoming increasingly important as it is closely linked to the expectations of battery technology. The next evolutionary stage of sustainable mobility always develops initially at the extremes – whether in racing or the aviation sector. The interplay of intelligent digital technologies and novel materials, as well as their combinations and compositions that rely on silicon and other alternatives to active materials, create the basis for this. The next stage of electrification has already begun.
Dr. Daniela Werlich, CTO at CUSTOMCELLS