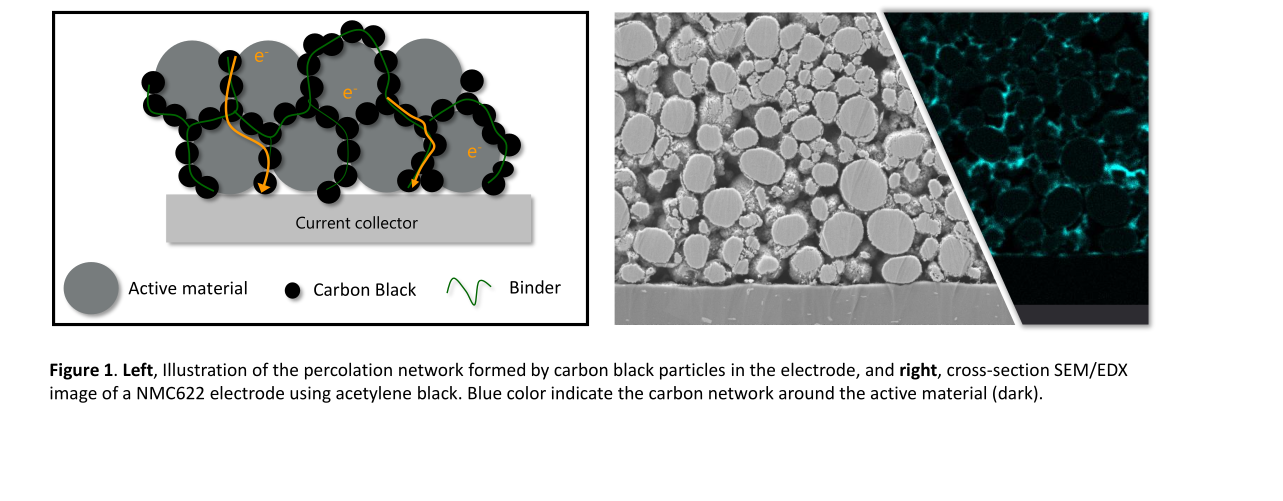
The Cell, the Battery and Battery Management societies are facing numerous challenges. These challenges span many different disciplines, technologies and applications. Experience indicates that many battery pack and battery management designers and manufacturers overlook the key challenges of material selection for assembly, placement and chemical and thermal protection. This is to be expected as information available, classically only concentrates on key characteristics like Thermal Conductivity. Although these key characteristics are considerably important, they do not always deliver the desired need or result.
Before going into details about the material selection, we first investigate the generic characteristics of the main chemistries involved. In Table 1 we see for example that Silicones are soft, Epoxies rigid and Polyurethanes somewhere in the middle. Plus, the reparability; adhesion where the more adhesion the more difficult it is to remove.
In Table 2, we highlight the typical characteristics that are influenced when modifying chemistries to meet certain functional challenges.
Table1

Table2

When selecting a material it is taken that ease of processing is inevitable and that technical properties remain unchanged and in many cases it may be so. However how does this material fit within the existing processing environment? How easy is it to integrate and scale-up and will this have an effect on the technical properties? Using the simple formula Processability, can help to achieve the desired technical properties without compromise.
First material flow, the thixotropic properties of the material; how easy it will spread. The mix ratio, an important criteria for the equipment suppliers. 1:1 mix ratio is considered desirable but could create other bottle-necks typically if fillers are present in both A & B components, resulting in higher equipment costs and maintenance.
Reactivity range, the speed at which the material will for example begin to gel. A potential major bottle-neck and a common misperception “what is used for prototyping is not always best suited for mass-production”. These characteristics are off-set by Thermal conductivity, namely solid contents or filler type and volume. Also the viscosity ratio is always important especially for dispensing and equipment suppliers.

A major consideration for battery pack design is thermal conductivity; how to remove the heat from the cells or modules. This should never be confused with Thermal propagation or runaway. However, Thermal conductivity does play a key role in heat dissipation. In simple terms, thermal conductivity is the heat path between two points. It is the transfer of this heat from point A. (i.e., a cell or module) to point B. (i.e., the cooling plate or channel). The main concern of thermal conductivity alone is it only refers to the heat transfer and the link or path and the bulk material value. The reliable dissipation of heat and excess heat is essential for the stability and longevity of the system. For heat dissipation to be optimised Density, Wettability and Compressibility are all key factors. Here we should also include adhesion, in many cases a critical component for thermal conductivity but also a growing concern as battery packs become integral to the vehicle structural integrity and face greater mechanical stress. These characteristics are off-set by the layer thickness and here we can also add substrate type and mechanical stress.

E-mobility vehicle development strives to increase range while reducing total weight. The system weight is therefore of critical importance in battery development and yet surprisingly material density is sometime overlooked. Thermal conductive Gapfill materials are the largest contributor to weight. For every 1Ltr of 3.5W·mk material, with a typical density of between 3 to 3.5g/cm3, adds approximately 3.5kg of weight. Considering a modest size battery pack employs more than 5Ltrs of Gapfill material, this adds 15 to 20kg of unwanted weight. We can discuss low density materials, employing exotic filler types but cost is currently unrealistic. Some of these fillers increase the viscosity or changes thixotropic or more drastically the hydrophilic behaviour. Using materials with higher adhesion and better flowability it is possible to improve heat dissipation and reduce weight by employing lower thermal conductive (density) materials.


Taking these considerations a step further, base material chemistries can be compared. Graph 1 represents key functionalities required. From the top clockwise:
- Thermal Conductivity from Low 1W·mk up to currently 3.5W·mk and possibly higher.
- Mechanical Adaptability from Low to high, elastomeric, hardness, dampening and cycling behaviour.
- Flame Retardant properties from weak to good. Capability vrs Permanent operating temperature.
- Temperature stability from low to high. Long term thermal stability and cycling behaviour
- Adhesion from weak to good. Chemistry, substrates, environment.
- Impregnation from weak to good. Application and environment where the material will be employed.
- Insulation properties from Low to high. Dielectric strength, breakdown voltage.
- Reactivity Range from narrow to wide. Versatility to adjust the gelation speed.
As the graph shows, Silicone has excellent Thermal Conductivity, good insulation properties, UL and a high permanent operating temperature. For Polyurethane, due to the thixotropic behaviour of the material with large filler content, Thermal conductivity is lower and the natural temperature boundary of the chemistry, a lower Thermal stability. However for Mechanical strength (namely adhesion) and the reactivity time, the speed the material can or not gel, Polyurethane has more range capability. Critical for designs requiring mechanical reliability.
In Graph 2, the same data for Silicone versus Epoxy. For battery pack designs due to the potential rigidness or crack potential, epoxy is not considered. For applications such as motors, then epoxy is of interest, especially impregnation and adhesion properties.
WEVO Chemie GMBH is a family owned, independent company headquartered in the German automotive manufacturing hub, Stuttgart. Their global presence supports the major electrical and electronic societies. Since producing their first polyurethane-based electrical casting resins in 1978, the company has continued to develop and supply a range of solutions for OEM’s and Tiers in the automotive, home, engineering and energy sectors. They have taken the lead as a supplier of encapsulation solutions for automotive sensors and in 2007 supplied their first resin solution for an EV battery system, which is still running today.
Author: Terence Kearns and Jochen Frank